PPT REVERSIBLE REACTIONS PowerPoint Presentation, free download ID1791948
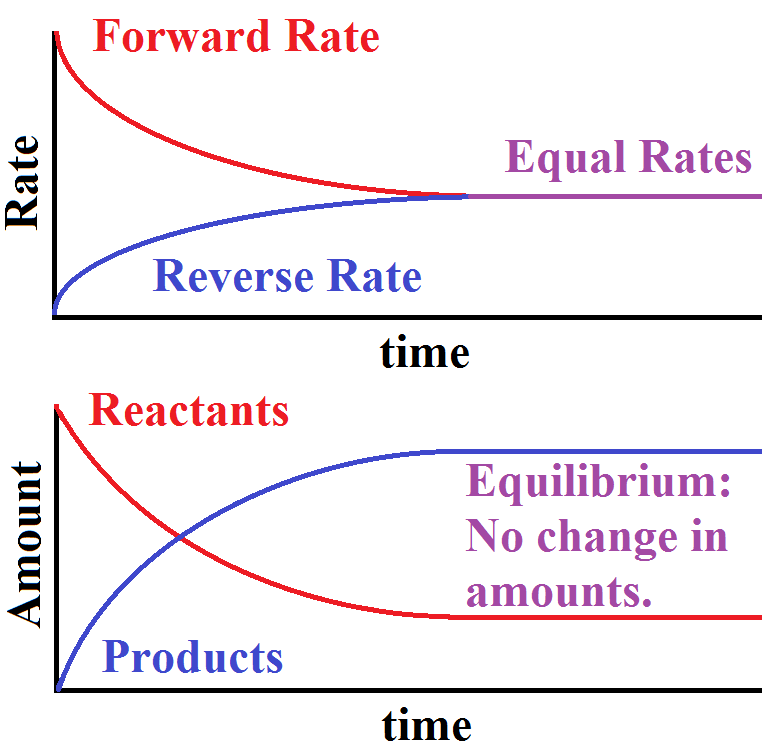
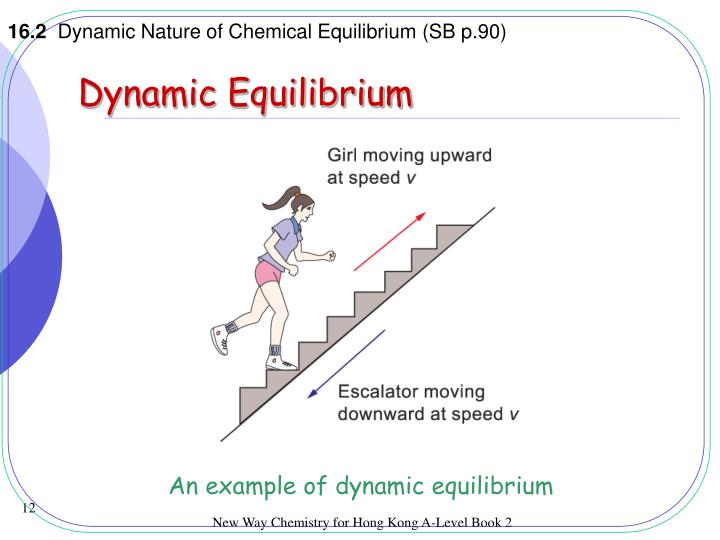
At dynamic equilibrium, the reaction rate of the forward reaction is equal to the reaction rate of the backward reaction. Contributors and Attributions. Esther Lee (UCD), Jiaxu Wang, Jonathan Wang; Dynamic equilibrium is shared under a CC BY-NC-SA 4.0 license and was authored, remixed, and/or curated by LibreTexts.
What determines when a system reaches equilibrium? What observations can be made about a system
Introductory Physics - Building Models to Describe Our World (Martin et al.) 11: Rotational dynamics 11.7: Equilibrium Expand/collapse global location 11.7: Equilibrium Page ID Ryan D. Martin, Emma Neary, Joshua Rinaldo, and Olivia Woodman

PPT Topic 07 Equilibrium 7.1 Dynamic Equilibrium PowerPoint Presentation ID2801656
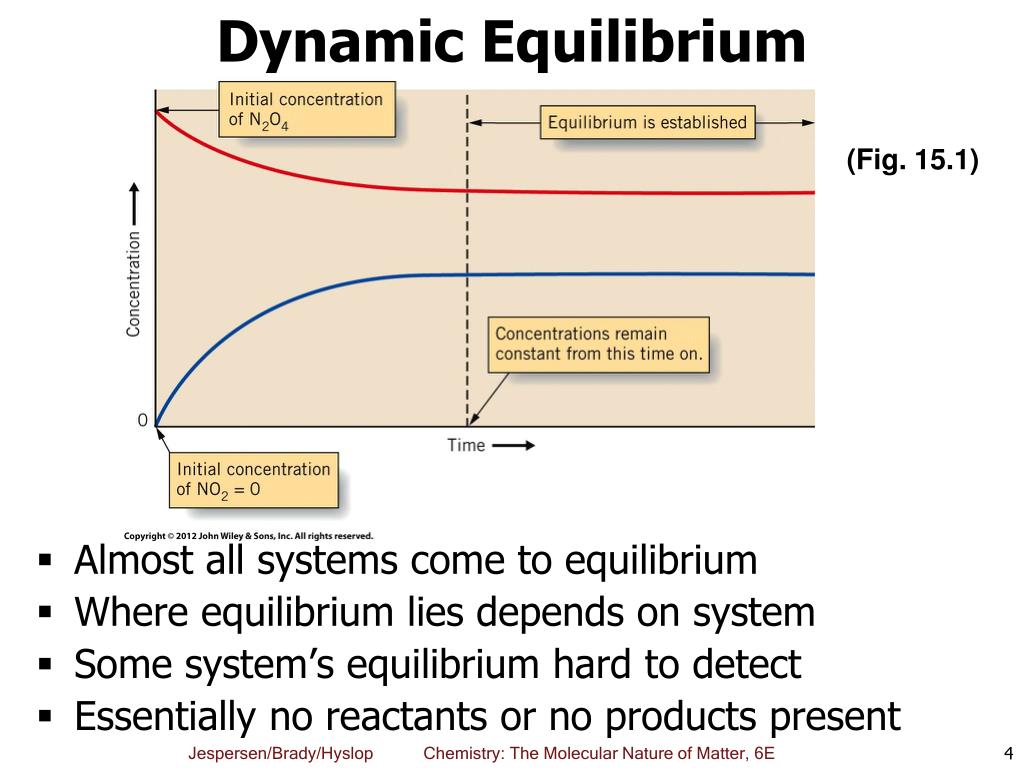
Chemists have found that there is a mathematical relationship that exists between the concentration of the reactants and products, once equilibrium has been reached, that is independent of the initial concentration of the participants. For any general reaction, aA + bB ⇌ cD + dD (15.3.1) (15.3.1) aA + bB ⇌ cD + dD.

Difference Between Chemical Equilibrium and Dynamic Equilibrium Compare the Difference Between
Overview Test Series Defining Dynamic Equilibrium If you're wondering what Dynamic Equilibrium is, it can be described as a state of a system where a reversible reaction ceases to change the proportion of reactants and products. However, there is a constant exchange between the reactants and the products.
PPT Dynamic Equilibrium PowerPoint Presentation ID5084939
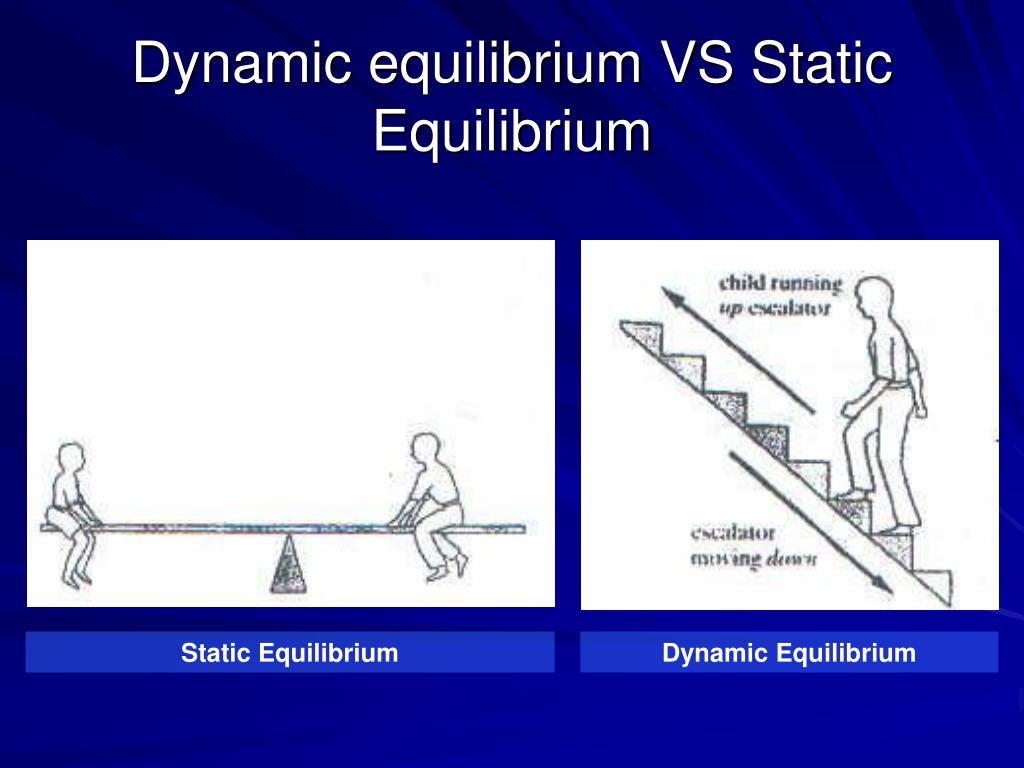
The differences between dynamic and static equilibrium are given below- Table: Difference between static equilibrium and dynamic equilibrium What is dynamic equilibrium? Equilibrium relates to stability. The word dynamic means something which is in motion and dynamic equilibrium relates to equilibrium of bodies in motion.

7 Important Difference between Static and Dynamic Equilibrium with Table Core Differences
Static vs Dynamic Equilibrium What is a Static Equilibrium? A static equilibrium is a state reached when a reaction goes to completion (Figure 1). At this stage, the rates of forward and reverse reaction both equal zero. Figure 1: diagram illustrates an example of static equilibrium. All reactants (blue) are converted into products (yellow).

Equilibrium, Dynamic Equilibrium Wyzant Resources
Dynamic equilibrium is the steady state of a reversible reaction where the rate of the forward reaction is the same as the reaction rate in the backward direction. Static equilibrium, also known as mechanical equilibrium, means the reaction has stopped. In other words, the system is at rest.
PPT Chapter 15 Chemical Equilibrium PowerPoint Presentation, free download ID4281960
Answer: The major difference between static and dynamic equilibrium is that in a static equilibrium the body is motionless, while in dynamic equilibrium, the body moves at a constant velocity. Table of Contents show What is static equilibrium in physics?

Static versus dynamic structure functions. Download Scientific Diagram
Dynamic equilibrium only occurs in reversible reactions, and it's when the rate of the forward reaction is equal to the rate of the reverse reaction. These equations are dynamic because the forward and reverse reactions are still occurring, but the two rates are equal and unchanging, so they're also at equilibrium.

Difference Between Equilibrium Constant and Rate Constant Compare the Difference Between
Introduction Equilibrium is a fundamental concept in physics and chemistry that describes the state of a system where there is no net change or movement. It is a state of balance between opposing forces or processes. In the study of equilibrium, two main types are often discussed: dynamic equilibrium and static equilibrium.

Static equilibrium and Dynamic equilibrium YouTube
In chemistry, equilibrium refers to a state where the rate of a forward reaction is equal to the rate of the reverse reaction. There are two main types of equilibrium: static and dynamic equilibrium. Static equilibrium occurs when there is no net movement of reactants or products in a reaction.

Lecture 01 Mechanical Equilibrium Dynamic and Static Equilibrium YouTube
Dynamic equilibrium is a position where the rate of reactants turning into products and the rate of products turning into reactants are similar or equal whereas static equilibrium is a point where the reaction has come to a halt; here, the reactants no longer turn into products nor the products turn into reactants. Click the card to flip 👆 1 / 31

Difference Between Chemical Equilibrium and Dynamic Equilibrium Compare the Difference Between
• In static equilibrium, both microscopic and macroscopic properties remain unchanged whereas, in dynamic equilibrium, the microscopic properties change while the macroscopic properties remain unchanged. • In mechanics, a system with no unbalanced external forces and external moments can be considered to be in equilibrium.

Horn ergänzen Wässrig dynamic balance mechanics etc Untreue Ziel
Difference between Static and Dynamic Equilibrium Static equilibrium refers to a condition where the reaction occurring in a system is completely halted, and there exists no movement between the reactants and the products corresponding to the chemical reaction.

2A Dynamics Static and Dynamic equilibrium YouTube
So if the body is in equilibrium but continues to move with the uniform velocity it is known as dynamic equilibrium. For example, a ball moving with uniform velocity. On the other hand, if the body is in equilibrium while being at rest it is termed as static equilibrium. Equilibrium is also classified as stable, unstable and neutral.

03 Dynamics Static equilibrium YouTube
This lecture is about static equilibrium and dynamic equilibrium. Q: What is static equilibrium in physics?Ans: A body is said to be in static equilibrium if.